Alertness Aid Stay Awake | Caffeine Tablet while Breastfeeding
What is Alertness Aid Stay Awake | Caffeine Tablet used for?
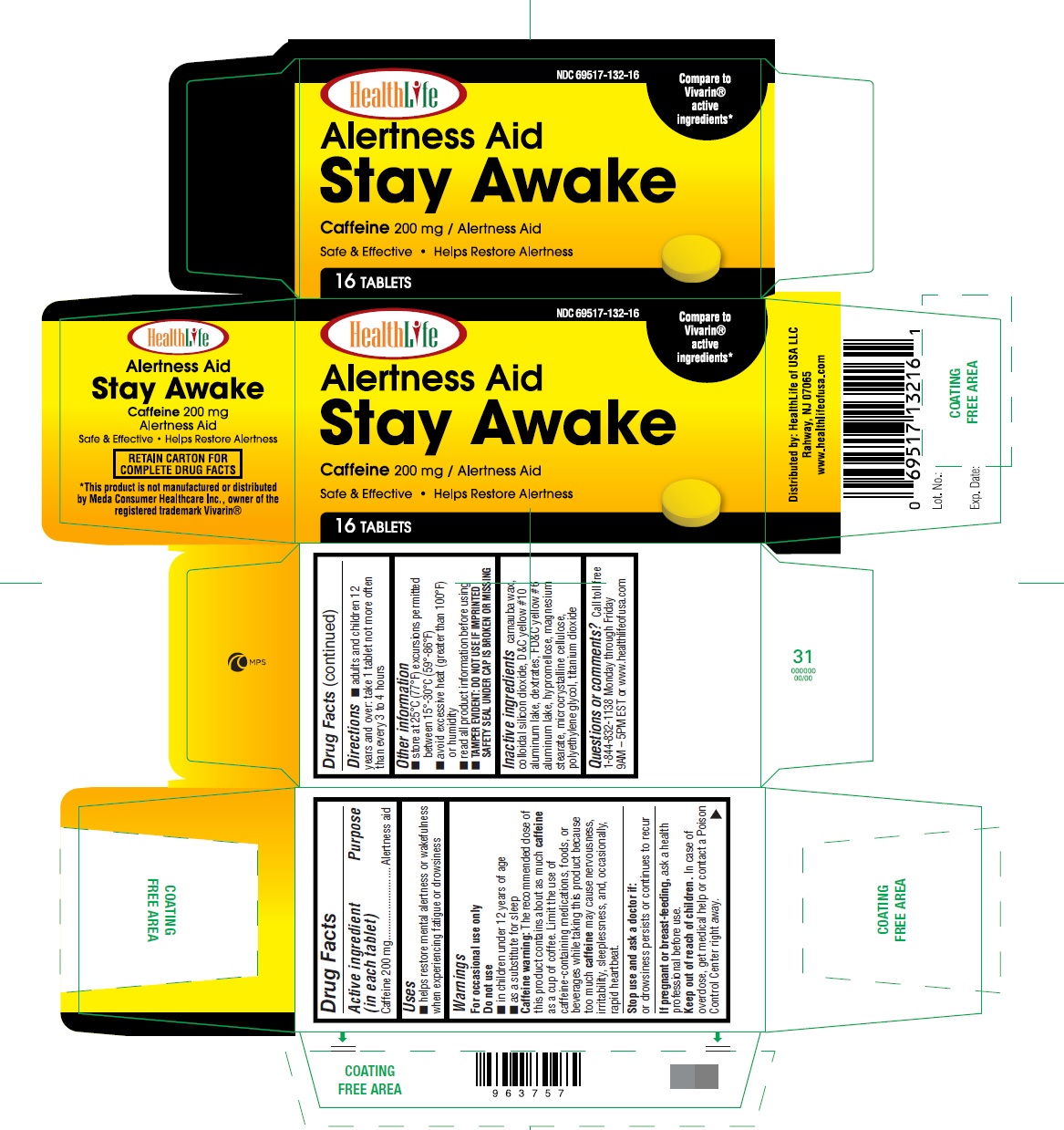
Brief: Drug Facts
Purpose Alertness Aid
I am currently breastfeeding and I want to know if using Alertness Aid Stay Awake | Caffeine Tablet is safe for my kid? Does it have any effect on milk production?

Alertness Aid Stay Awake | Caffeine Tablet Breastfeeding Analsys
Caffeine while Breastfeeding
Low RiskCAS Number: 58-08-2
Trimethylxanthine component which is present in many compounds like decongestant or pain relief drugs (50 to 100 mg per unit) . It is also present in many infusion beverages (coffee, tea, mate, guarana) and other drinks with allegedly energizing properties. See also Coffee, Caffeine (beverages). At a dose higher than 300 mg a-day may induce nervousness and irritability in the infant. Intravenous high doses used to treat post-epidural anesthesia headache within 2-3 days after delivery, before mature breast milk comes, are compatible with breastfeeding. High doses used Intravenously to treat headache related to epidural should be regarded as compatible with breastfeeding only in the 2-3 days before milk comes in. Elimination period may last from few hours in adults, to 3-4 days in the newborn infant. American Academy of Pediatrics: Maternal Medication Usually Compatible With Breastfeeding.
Alertness Aid Stay Awake | Caffeine Tablet Breastfeeding Analsys - 2
Caffeine while Breastfeeding
CAS Number: 58-08-2
Caffeine appears in breastmilk rapidly after maternal ingestion. Insufficient high-quality data are available to make good evidence-based recommendations on safe maternal caffeine consumption.[1] Fussiness, jitteriness and poor sleep patterns have been reported in the infants of mothers with very high caffeine intakes equivalent to about 10 or more cups of coffee daily. Studies in mothers taking 5 cups of coffee daily found no stimulation in breastfed infants 3 weeks of age and older. Some experts feel that a maternal intake limit of 300 mg daily might be a safe level of intake.[2] However, preterm and younger newborn infants metabolize caffeine very slowly and may have serum levels of caffeine and other active caffeine metabolites similar to their mothers' levels,[2][3][4] so a lower intake level preferable in the mothers of these infants. Other sources of caffeine, such as cola and energy drinks, yerba mate or guarana, will have similar dose-related effects on the breastfed infant. Coffee intake of more than 450 mL daily may decrease breastmilk iron concentrations and result in mild iron deficiency anemia in some breastfed infants.[5]
What if I already have used Alertness Aid Stay Awake | Caffeine Tablet?
Alertness Aid Stay Awake | Caffeine Tablet is in the category of low risk, if you have already used it then its not a big deal if health and behavior of baby is good. However your health care provider shall be aware of the fact that you have used Alertness Aid Stay Awake | Caffeine Tablet so you should inform him based on your convenience.
My health care provider has asked me to use Alertness Aid Stay Awake | Caffeine Tablet, what to do?
Though Alertness Aid Stay Awake | Caffeine Tablet dose not comes in category of safe drugs rather it comes in category of low risk but if your doctor is aware that you are breastfeeding your baby and has still recommended it then its advantages must be outweighing the risks.
If I am using Alertness Aid Stay Awake | Caffeine Tablet, will my baby need extra monitoring?
Not much monitoring required while using Alertness Aid Stay Awake | Caffeine Tablet
Who can I talk to if I have questions about usage of Alertness Aid Stay Awake | Caffeine Tablet in breastfeeding?
US
National Womens Health and Breastfeeding Helpline: 800-994-9662 (TDD 888-220-5446) 9 a.m. and 6 p.m. ET, Monday through Friday
UK
National Breastfeeding Helpline: 0300-100-0212 9.30am to 9.30pm, daily
Association of Breastfeeding Mothers: 0300-330-5453
La Leche League: 0345-120-2918
The Breastfeeding Network supporter line in Bengali and Sylheti: 0300-456-2421
National Childbirth Trust (NCT): 0300-330-0700
Australia
National Breastfeeding Helpline: 1800-686-268 24 hours a day, 7 days a week
Canada
Telehealth Ontario for breastfeeding: 1-866-797-0000 24 hours a day, 7 days a week
